
What are some of the side effects from using Medtronic Infuse Bone Graft? Two individual reports that came out in 2013 found that Infuse is no more beneficial than regular spinal surgery and that the product can cause serious medical issues. It is alleged that Medtronic paid 13 doctors and 2 corporations over $210 million to co-author a series of papers trying to advance and heighten the product in the medical community. However, what has not been approved is the off label use of the infuse bone stimulator in the cervical spine. In 2002 the FDA approved Medtronic’s bone graft to aid in the treatment of degenerative disk disease and for limited use in the lower back area. Infuse is a biologic agent that has been implanted in over 1 million people to replace spinal discs as well as generate more bone growth in the spinal area. One of the latest issues facing this Fortune 500 company are lawsuits filed by over 1,000 people regarding the bone morphogenetic protein (BMP-2) product known as Infuse, which is used in spinal surgery. Gform.Minnesota-based Medtronic, the fourth largest medical device company in the world, is no stranger to lawsuits. Δ document.getElementById( "ak_js_2" ).setAttribute( "value", ( new Date() ).getTime() ) We tell you about cash you can claim every week!ĬAPTCHAPhoneThis field is for validation purposes and should be left unchanged.

If true, this can have legal implications. Off-label purposes are those other than what has been approved by the U.S. Additionally, it has been alleged that Medtronic promoted Infuse for “off-label” purposes. The spine is rich in delicate, non-healing spinal tissue wrapped in nerve-dense muscles and connective tissues. In many cases spinal surgery can cause more harm than good. In some cases, surgeons have been unable to fix the problem. This alleged overgrowth of bone can damage and compress nerves in the spine. Many of these involve the overgrowth of bone. In Infuse bone graft lawsuits, the procedure has been linked to a number of very serious complications. This means that spinal fusions with Infuse don’t require a second procedure to harvest redundant bone from elsewhere in the body. The big benefit of the Infuse system is that the gel is made of a small tissue sample from the patient combined with synthetic proteins and other compounds. The cage supports the two bones of the spine and the gel encourages bone to grow. The Infuse system consists of two parts: a metallic cage and bioengineered gel.
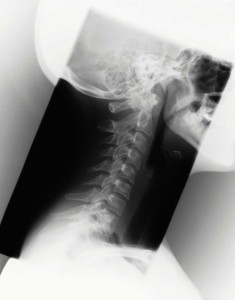
Infuse was supposed to be a better way of doing this. Once in place, the bone tissue joins the two bones into a single bone and supports them, reducing the pressure on the damaged disc. In traditional spinal fusions, surgeons harvest bone from elsewhere in the body and graft it to the spine. As some people age, wear and tear to these discs causes them to fail, leading to back pain and damage to the spine. Spinal discs are cushions of connective tissue between the individual backbones, or vertebrae. Medtronic’s Infuse bone graft system is designed to improve spinal fusion surgery, a procedure intended to help people with damage to their spinal discs.

More than 1,000 patients have filed Medtronic Infuse bone graft lawsuits alleging the medical implant can be dangerous and cause crippling side effects.
